Schoffelen & Plasqui explained the various EE components in a respiration chamber and sources of error in indirect calorimetry in their 2017 paper: “Classical experiments in whole-body metabolism: open-circuit respirometry—diluted flow chamber, hood, or facemask systems”.
Human energy expenditure (EE) is not constant and varies with the time of day, activity level, meal digestion, and other factors. The momentary EE measured at each instant in time is the same as the sum of the EE components. Even though they are never evaluated separately, certain of these EE components can be identified individually during specific times, such as during sleep, rest, exercise, meals, or over the course of a 24-hour period (Fig. 6).
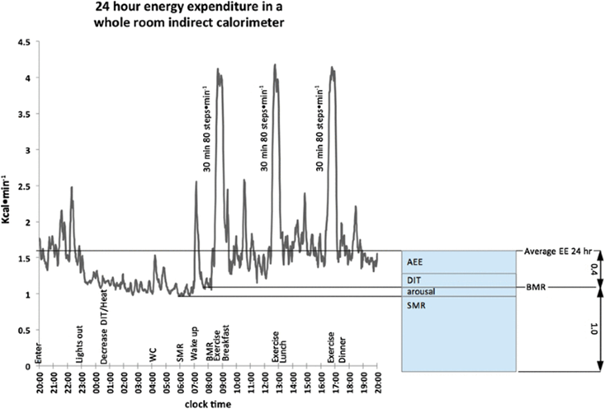
Fig. 6
Several EE components were evaluated during a 24-hour stay in a breathing chamber. The patient enters at 20:00 and remains for 24 hours in accordance with the method, which is described above the X-axis clock time. EE decreases over the night as DIT and body heat do. During the last stages of sleep, SMR is calculated. The BMR measurement was made possible by a brief time of waking sleep on the bed after awakening, tooth brushing, and showering. Throughout the day, three 30-minute walking sessions at an 80-step-per-minute rate were accomplished, each of which was followed by a meal. The result of diet- and activity-related thermogenesis, the average EE over a 24-hour period is 40% higher than BMR. A physical activity factor of 1.4 was noted for a stay inside the restricted space of a breathing chamber (Westerterp and Kester 2003)
These specific EE components don’t just start and stop. Each component will have a distinct response that may begin after the interval or may continue to work for a number of minutes to several hours or even days after the time has ended. Due to these time-course effects, the technique used greatly affects the capacity to examine the repeatability of EE components and compare results to those published in the literature. For example, EE values of sleep (SMR), defined as the lowest recorded EE across a 3-hour period at night, often between 3:00 and 6:00 in the morning, will differ dramatically from EE values of sleep (overnight) for an 8-hour period (23:00-07:00). The same is true for resting metabolic rate (RMR), which provides information on EE at rest, and basal metabolic rate (BMR), which has a more stringent definition than RMR. BMR is measured in the early morning after an overnight fast, when awake and in a supine position, in a thermoneutral atmosphere. RMR additionally designates an EE level of rest, typically following several hours of inactivity. The location, the time of day, and other factors could change, though. RMR would be significantly higher than BMR, for example, if the patient was active in the morning and RMR was measured several hours after lunch while the subject was sedentary.
Figure 6 depicts an example of a day’s worth of EE measurements made in a respiration chamber, showing the major components SMR, BMR, and average EE throughout 24 hours. Due to the fact that DIT and AEE occur at the same time each day and cannot be distinguished from one another without measuring additional parameters, they are difficult to separate from one another. This experiment, which depicts the subject using the lavatory in the middle of the night to highlight how SMR may be interrupted if activity is not taken into consideration, even uncovers a typical example of biological variation in a strictly controlled environment.
Other variables, such as the seasons or climate, health issues, high activity levels, nutrition, travel, age, gender, body type, hereditary characteristics, frequency of visits, etc., can also have an impact on biological variance.
Since studies frequently compare the results of one group after an intervention, or imposed variation, to those of a control group, understanding biological variation is essential for study design.
In this variety of EE components, accuracy and measurement interval in relation to EE level are important considerations. An indirect room calorimeter will have a floor noise level determined by the calorimeter’s volume and analysis accuracy as well as a base accuracy level that is virtually constant and irrespective of the quantity of EE. It is a variable that can change over the course of the measurement, becoming larger for shorter periods of time and smaller for longer ones. The source of a second component with a similar floor noise level is the throughput flow and its analysis. When combined together, this noise level is constant across time and gets louder over time. With the exception of the fact that in actuality different degrees of EE may have varied flow rates, both are unaffected by the actual level of EE. In a medium-yet-constant flow condition, the floor-level accuracy is almost constant, and the absolute SD for discoveries is comparable for low-level EE and high-level EE. Alternatively, high levels of EE can be monitored over a shorter time period with the same accuracy as low levels over a longer time period. High levels of EE will therefore have less accuracy than low levels of EE. This aspect needs to be taken into account while choosing the measurement interval for different EE components. For instance, it sometimes takes many hours to determine the lowest EE level of sleep, but it just takes 20 minutes to determine the higher levels.
Sources of error in indirect calorimetry
Human indirect calorimetry contains many sources of inaccuracy (McLean and Tobin 1987). To categorize a non-exhaustive sample of typical sources of errors, theoretical, system (random and systematical), and practical errors might be used.
The choice of formula and assumption is where the majority of theoretical errors originate. Typical presumptions include the absence of leaks, thorough mixing of all gas volumes before evaluation, the ideality of all gasses, and the unaltered passage of inert gas through the calorimeter (Haldane correction). Because it is based on the substrate composition (Livesey and Elia 1988), which is thought to be the same as that consumed by the individuals in question, the formula used to convert gas-exchange data to EE (Weir 1949; Brouwer 1957) is a typical example.
The validation tests must also be able to simulate the biological application in this case. For instance, while studying a breath-by-breath (BxB) system with alcohol combustion in diluted flow mode, it should be noted that the BxB measurement is a different mode from the diluted flow (ventilated hood) measurement.
The technical design of the calorimeter results in random or systematic system errors. The former average out over time, whereas the latter could eventually lead to a higher cumulative error.
Examples include calibration accuracy, measurement noise and drift, digitalization characteristics, cleaning up system debris and flushing the system, calibration noise and drift, and inappropriate validation approaches (i.e., not suitable for the biological application). One biological example is the effect of CO2 diffusion through the skin, which accounts for a + 1-2% difference in measured CO2 between vented hood or facemask calorimetry and whole-room calorimetry.
Practical errors result from the calorimeter’s operation, and they can be brought on by either human error or deficiencies in the surrounding conditions or available resources. Examples include disregarding protocol, such as incorrectly calorimeter sealing or other causes of leakage, or incorrect calorimeter flow setting, resulting in an abnormally large or excessively little dilution flow. Local factors may have an impact on temperature, ambient air composition, and necessary supplies, such as calibration gasses that may have an erroneous value or certificate correctness.
As usual sources of inaccuracy, push vs. pull systems, leakage, and tainted incoming air have all been mentioned. The volume of air that is present as well as compositional changes brought on by a single individual are both counted by an indirect calorimeter. There may be absolutely no air from outside sources or from the topic itself.
It’s possible that a decline in the availability of specialized knowledge occurred at the numerous locations using indirect calorimetry as a result of the proliferation of small indirect calorimeters (metabolic carts), along with a concurrent decrease in the amount of work required to conduct studies. There are times when an indirect calorimeter can be compared to a mysterious black box that measures EE in people. However, it is still a delicate measurement. For instance, a ventilated hood with a flow of 40 l per minute is frequently used to define a ventilated hood’s acceptable accuracy, which may be 5%, or 10 ml per minute of oxygen consumption, for BMR measurements. However, this baseline requirement of merely 5% biological precision calls for a technological accuracy of 0.025% (or 10 40,000 1). It can be confusing and results in an underestimation of technical effort because of the dramatic difference between biological and technical accuracy needs.
The technical effort necessary will increase to the point where personnel must acquire the essential skills and experience before achieving even the most basic operating status because whole-room calorimetry errors are becoming more sensitive.
Keep in mind that experience can sometimes have detrimental consequences because practices and expertise change as technology requirements do. It is nevertheless possible to miss an apparatus’s drift even with tried-and-true operating procedures since it constantly stays within the previous “approved” error range. A calorimeter that needs to operate with a 5% absolute error, which is equal to 1 2%, would serve as an example (mean SD). The mean SD may now pass inspection if SD dropped from 2 to 0.5%, but a change in the procedures should actually keep it within 1 0.5%, or within 2%, rather than the maximum error range of 5%.
Related products
Whole body room calorimeters
The Room Calorimeter offers the highest validated accuracy and reproducibility in the market. Designed on a system level out of the highest quality components, this is the gold standard for energy expenditure studies of any kind; 24-hr energy expenditure, high intensity exercise testing and many more. Validated and applied in 100’s of research studies.
Omnical
The Omnical is the market’s most versatile and accurate indirect calorimeter for research. It allows users to conduct investigations in a variety of study sectors using cutting-edge technology and top-of-the-line precision measurement devices. The system is designed to monitor energy metabolism with great precision, ranging from resting metabolic rate (RMR) through sports performance testing (e.g. VO2max tests).
How can we help you with your research?
Maastricht Instruments creates equipment in the field for indirect calorimetry measurements. We provide support for studies, research and measurements alongside our indirect calorimetry products.
Consult us about our indirect calorimetry metabolic cart, whole room calorimeter systems or accelerometry add-ons. Please contact us or find more information on our information pages.
Reference
Schoffelen, P.F.M., Plasqui, G. Classical experiments in whole-body metabolism: open-circuit respirometry—diluted flow chamber, hood, or facemask systems. Eur J Appl Physiol 118, 33–49 (2018). https://doi.org/10.1007/s00421-017-3735-5
